My CIDP log of CIDP hitting 80 years-old 2026, the end game: No drugs possible? + Aging with CIDP Progression + Battle for VYVGART / efgartigimod alfa after my own NCS analysis:
January 2026: I hit aged 80 years old and I am in a quandary with no options available for relief from CIDP progression after minimally 15 years of CIDP. December 15, 2025: Year-end appointment with my CIDP doctor is depressing. The CIDP Clinic Nerve Conduction Studies NCS confirm I am worse and talk of me ending in a wheelchair this year 2026 prevails with no therapy available. All historic symptoms are worsening, notably the top ten in the last year increases in:
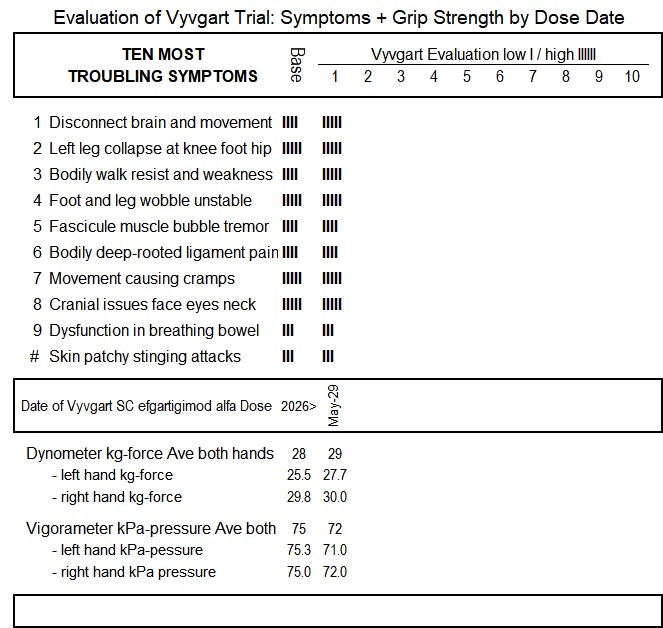
• disconnect between my brain and movement
• left leg collapsing at knee, foot and hip
• bodily walking resistance and weakness
• foot and leg wobble / walking instability
• fasciculation (muscle bubbling, waves, vibration and tremor)
• deep-rooted pain in ligaments everywhere
• any beyond average movement causing cramping
• greatly increased cranial issues and neck
• autonomic dysfunction in breathing (breathing hiccups) and bowel movement
• new weird surface skin patchy very painful stinging attacks
Still apparently no treatment options (such as Rituximab) as repeated blood tests are negative for nodal antibodies anti-neurofascin-155 (NF155) as well as Neurofascin-140, NF155, NF186, ONTN1 and CASPRI as well as Immunofixation C Reactive Protein. All results were negative.
This watershed year 2026, I also sensibly started my own analysis of my nerve conduction studies NCS and have discovered wide negative differences between what I have concluded from the data versus what I had thought prior to my analysis So I singlehandedly early 2026 searched for and have learned about a new CIDP drug in Europe by Argenx branded Vyvgart SC which I am pursuing.† .
Summary Log My CIDP 2026 Big Challenges with No Treatment?
CIDP Chronic Inflammatory Demyelinating Polyneuropathy the End-Game at aged 80
» February 19, 2026: I hire an an EMG Technologist with 30-years experience and her daughter a whiz-kid data analyst to assist me with my own analysis of the many nerve conduction studies I have had since 2002.
» March 16, 2026: Results of my own NCS start coming in. Looks like I was in deterioration free-fall December 13, 2022 when the official CIDP Clinic report read my "nerve conduction studies are stable to better" and "Improved".
» March 30, 2026: I am mortified by my NCS analysis findings. I take action to find new treatment options, firstly by trying to contact Argenx directly (in Europe and North America) who launched a new drug VYVGART Hytrulo / efgartigimod alfa.† As I previously turned allergic to IgG in October 2020, I have assumed that I would also be allergic to the VYVGART Hytrulo but now I am not so sure. I would like to urgently determine whether I would be allergic.
» April 01, 2026: I get a call from the Canadian manager of Argenx operations. I am told Vyvgart SC was approved in Canada as a treatment for CIDP November 05, 2025 but is only in the pipeline for funding under OHIP (which is the Ontario Canada Provincial medical insurance plan). Vyvgart SC is administered weekly at a cost of CAD$20,700 each jab!
» April 01, 2026: I have told Argencx that I am willing to pay for Vyvgart SC / efgartigimod alfa for a defined short period if this will prove I am not allergic. I am wondering if I will be allergic and also whether I will be susceptible to respiratory infections (a major side-effect). I am also hopeful that I will not actually end-up paying the whole amount (which I really cannot afford)
» April 16, 2026: I am having a hard time getting any traction on my bid to get a trial of Vyvgart SC in Canada even though I was allergic to IgG and my NCS showing a worrisome downward trend. I ask myself "Has the Canadian medical system, and CIDP Clinic let me down again?"
» April 20, 2026: I consult a neurologist in Holland on Vyvgart access there, and discover even in Holland this is problematic due to its high cost.
» April 30, 2026: I consult with a different doctor at my Toronto CIDP Clinic, who finally enrolls me in the Vyvgart SC program. Once accepted I will be able to start the therapy at my own expense until the government funding is approved (expected "anytime now" although most likely September 2026). Big financial decision ahead for me - do I invest in my health now or take a big risk and wait for the government funding? At this time, I am thinking I must proceed.
» May 15, 2026: I am at breaking point with my left leg collapsing several times each day. Having followed-up with the CIDP Clinic and Argenx rep, but still have not been able to expedite the Vyvgart SC first jab.
» May 22, 2026: I have early good news on funding but still waiting for the date for the first dose injection to be set.
» May 28, 2026: Amazing news from Argenx who have offered to fully fund six months of Vyvgart SC infusions weekly starting May 29, 2026. I am totally flabbergasted! This could be life changing!!
† Argenx Vyvgart Hytrulo, Vyvgart SC (efgartigimod alfa and hyaluronidase-qvfc) works in CIDP patients by significantly reducing the levels of harmful Immunoglobulin G (IgG) antibodies in the blood. It is designed to target the underlying autoimmune cause of the disease by attaching to neonatal Fc receptors (FcRn), which prevents these antibodies from being recycled and encourages their degradation. Fc receptors or FcRn blockers bind to antibodies that are attached to infected cells or invading pathogens."Fc" stands for fragment crystallizable. By out competing endogenous IgG, efgartigimod alf prevents recycling and promotes lysosomal degradation of IgG, leading to lower IgG levels, without affecting IgG production.
I am planning to track my response to Vyvgart SC using my current May 2026 ten most troubling CIDP symptoms and grip-strength measures. I have prepared the template I will use as below as I await the receipt of the first dose:.
Also see:
> 2026 January: My Nerve Conduction Study 2026 Analysis
> 2013 July: Daily log of 2nd eight-months round of IVIg July 2013 - April 2014
> 2015 March: Log of IVIG impacts March-October 2015
> 2015 October: Log of Sub-Q trial SCIG of Hizentra) Oct 2015
> 2016 March: Log Maintenance Dose IVIg Therapy Mar 2016 though 2018
> 2018 January: Log of clinical trial of Panzyga IVIG Jan-Dec 2018
> 2018 December: Log of Sub-Q Ig Cuvitru Sub-Q Dec 2018 through Oct 2020
> 2020 November: Log transition off IgG after Adverse Reaction Nov 2020
> 2021 February: Log Going-Solo no CIDP Drugs Feb 18, 2021 - July 16, 2024
> 2024 July: Log CIDP vs Eagle Syndrome July 16, 2024 - Dec 2025
> 2026 January: Log CIDP hitting 80 years-old - the End Game from Jan 2026
> My summary log and findings on my CIDP infusions of IgG 2011-2020
Concepts for My CIDP log of CIDP hitting 80 years-old 2026, the end game: No drugs possible + Aging and CIDP: old-age, retired, New drugs for CIDP, allergic to IgG in Canada with CIDP and old, efgartigimod alfa for CIDP Chronic Inflammatory Demyelinating Polyneuropathy (CIDP)
